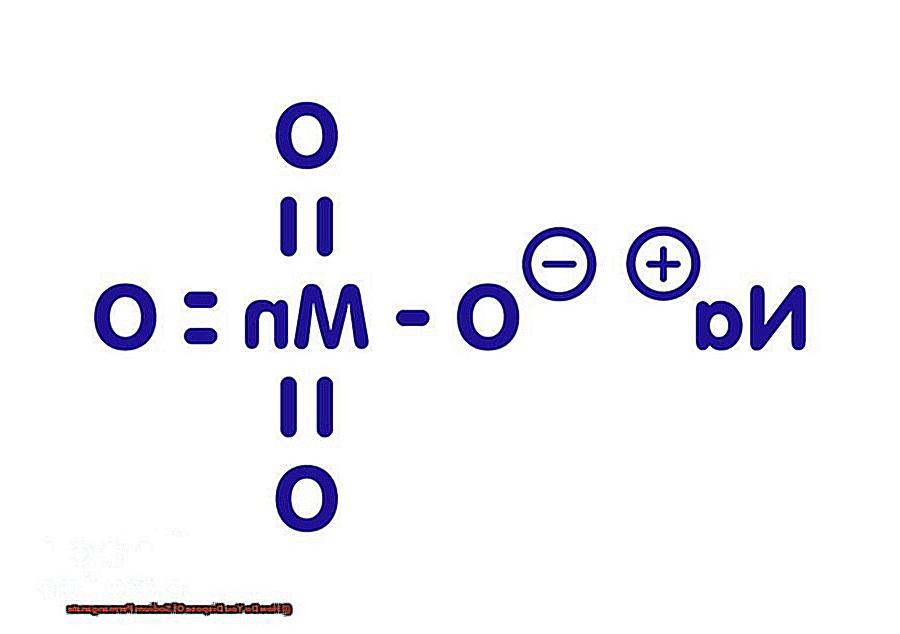
The vibrant purple chemical that’s a staple in many projects. But now that your project is complete, you’re faced with the task of disposing of it safely.
Don’t panic, we’ve got your back. In this post, we’ll guide you through the proper steps to bid farewell to this powerful substance without any hassle.
So put on your favorite playlist and join us as we uncover the secrets of responsible disposal.
How Do You Dispose Of Sodium Permanganate?
Contents
- 1 How Do You Dispose Of Sodium Permanganate?
- 2 The Hazards of Improper Disposal
- 3 Risks Associated with Sodium Permanganate
- 4 Safe Disposal Methods: Dilution with Water
- 5 Alternative Method: Using Reducing Agents
- 6 Precautions and Safety Measures for Disposing of Sodium Permanganate
- 7 Proper Disposal Procedures and Regulations
- 8 Conclusion
Sodium permanganate, a common chemical compound used in various industries, can be a beneficial substance when used properly. However, it can also pose a threat to our health and the environment if not disposed of correctly. As an expert in chemical safety, I’m here to share my insights and instructions on how to safely dispose of sodium permanganate.
First, it’s essential to understand the potential dangers of sodium permanganate. This chemical is corrosive and can cause skin and eye irritation, as well as release toxic fumes when mixed with other chemicals. It is also flammable and can lead to fires or explosions if not handled carefully.
So, how do we dispose of sodium permanganate safely? There are two common methods: dilution with water and neutralization with an acid solution. Let’s dive into each method in more detail.
Dilution with Water:
This method involves mixing sodium permanganate with a large amount of water to reduce its concentration and make it less harmful. Here’s how to do it safely:
Make sure you are in a well-ventilated area and wearing appropriate safety gear such as gloves and goggles.
Slowly add the sodium permanganate to a large container of water while stirring continuously.
Once diluted, the solution can then be safely disposed of down the drain.
Neutralization with Acid Solution:
Another option for disposing of sodium permanganate is by neutralizing it with an acid solution. Here’s how:
Add an acid solution, such as hydrochloric acid, slowly to the sodium permanganate until the color changes from deep purple to pale pink.
This color change indicates that the chemical has been neutralized.
Once neutralized, the solution can be safely disposed of according to local regulations.
It is crucial to note that sodium permanganate should never be mixed with other chemicals or disposed of down the drain without proper neutralization. This can lead to dangerous reactions and harm to the environment.
In some cases, if the amount of sodium permanganate is small, it can be safely disposed of in a sealed container with other household waste. However, it is always best to consult with professionals for proper disposal guidelines.
The Hazards of Improper Disposal
Sodium permanganate may sound harmless, but don’t be fooled by its name. This chemical compound, commonly used in water treatment and industrial processes, can pose serious hazards if disposed of improperly. As an expert on chemical safety, I have seen firsthand the consequences of improper disposal and the importance of following proper guidelines. In this section, we will explore the dangers of sodium permanganate and provide you with essential information to ensure safe disposal.
Contaminating Water Sources and Soil
One of the most significant hazards of improper disposal of sodium permanganate is its potential to contaminate water sources and soil. This compound is highly soluble in water, making it easily carried through runoff or groundwater. Once in the water supply, it can harm aquatic life and disrupt the delicate balance of ecosystems. Moreover, if spilled on soil, it can damage plant roots and affect soil fertility.
Releasing Toxic Fumes into the Air
Sodium permanganate can also release toxic fumes into the air if not disposed of properly. These fumes can irritate the eyes, nose, and throat, and prolonged exposure can lead to more severe respiratory issues. In enclosed spaces, these fumes can reach dangerous levels and pose a risk to anyone nearby. It is crucial to handle this chemical with caution and dispose of it accordingly.
Avoiding Legal Consequences
Aside from the harm it can cause to humans and the environment, improper disposal of sodium permanganate can also result in legal consequences. This chemical is classified as a hazardous waste by most countries due to its potential risks. If found guilty of improper disposal, individuals or companies may face fines or legal action.
Proper Disposal Methods
To avoid these hazards and consequences, proper disposal methods must be followed. The Environmental Protection Agency (EPA) recommends treating sodium permanganate as hazardous waste and disposing of it through a licensed waste management facility.
For small amounts, diluting it with water and neutralizing it with sodium thiosulfate before disposal is also a safe option. It is essential to check with local regulations for specific guidelines.
Risks Associated with Sodium Permanganate
Sodium permanganate may sound like a harmless household product, but this chemical compound carries significant risks that should not be taken lightly. From skin irritation to environmental contamination, improper handling and disposal can have severe consequences. As an expert on the topic, I want to educate you on the potential hazards of sodium permanganate and provide tips on how to dispose of it safely.
First and foremost, let’s address the risks associated with this highly reactive compound. Sodium permanganate is classified as an oxidizing agent, meaning it can release large amounts of oxygen when it comes into contact with other substances. In its solid form, it can cause irritation to the skin and eyes, and if inhaled, it can irritate the respiratory system. In its liquid form, it can cause severe burns. Yikes. Handling this chemical with care is crucial to avoid any harm to ourselves.
But it’s not just our health that is at risk. Improper disposal of sodium permanganate can lead to environmental contamination. The chemical can seep into soil and water sources, harming plants, animals, and even humans who come into contact with it. It can also react with other chemicals in the environment and produce toxic fumes. Plus, its high reactivity makes it a potential fire or explosion hazard.
To make matters worse, sodium permanganate is classified as hazardous waste by the EPA. This means that special handling and disposal procedures must be followed to ensure the safety of people and the environment. So what can we do to properly dispose of this chemical?
Firstly, if you’re working with sodium permanganate, make sure to wear protective gear such as gloves and goggles to avoid direct contact with your skin and eyes. When disposing of solid sodium permanganate, mix it with water before pouring it down the drain or flushing it down the toilet. This will dilute its concentration and reduce its potential harm to the environment. For liquid sodium permanganate, you must contact a hazardous waste disposal company for proper disposal.
Safe Disposal Methods: Dilution with Water
If you’ve ever used sodium permanganate for cleaning or laboratory experiments, you know that this chemical compound requires careful handling. But what about disposing of it? Improper disposal of sodium permanganate can have serious consequences, including skin irritation, environmental contamination, and even explosions. That’s why it’s crucial to know the proper methods for safely disposing of this chemical.
One of the most commonly recommended methods for disposing of sodium permanganate is dilution with water. This process involves mixing the chemical with a large amount of water, at least 10 times the amount of the chemical. This dilution reduces the concentration of sodium permanganate and neutralizes its effects, making it safe for disposal down the drain or in a chemical waste disposal system.
But why is diluting sodium permanganate with water so important? Simply put, this chemical can react violently if not properly diluted. Mixing it with water not only reduces its concentration but also helps to neutralize its oxidizing properties. Therefore, it is essential to use a large amount of water and ensure complete dilution before disposal.
This method is most suitable for small quantities of sodium permanganate, such as from laboratory experiments or household cleaning products. It is important to wear protective equipment like gloves and goggles when diluting sodium permanganate to avoid any direct contact with the chemical. Even when diluted, it can still cause skin irritation and damage.
In addition to using protective gear, it is crucial to handle the diluted mixture carefully and avoid splashing or spilling it. After disposal, make sure to thoroughly clean any containers or utensils used in the process to prevent any residual reactions.
However, this method may not be suitable for larger quantities of sodium permanganate or if there is no access to proper disposal facilities. In such cases, it is best to consult with a professional or local authorities for guidance on safe disposal methods.
Alternative Method: Using Reducing Agents
Sodium permanganate, a strong oxidizing agent, is notorious for its hazardous effects on both humans and the environment. Proper disposal of this chemical compound is crucial to prevent any accidents or harm. While dilution with water is the most commonly known method for safely disposing of sodium permanganate, there is an alternative method that utilizes reducing agents. In this article, we will guide you through the process of safely disposing of sodium permanganate using reducing agents such as sodium sulfite or ascorbic acid.
What are Reducing Agents?

Before we dive into the alternative method, let’s first understand what reducing agents are. In simple terms, reducing agents are substances that can donate electrons to another substance, causing it to undergo a reduction reaction. In the case of sodium permanganate, reducing agents can neutralize its oxidizing properties and make it safe for disposal.
Using Sodium Sulfite
One effective reducing agent for disposing of sodium permanganate is sodium sulfite. This readily available compound can be easily found in powdered form and can be mixed with water to create a solution. To dispose of sodium permanganate using this method, follow these steps:
- Dilute the sodium permanganate with water to make it easier to handle.
- Slowly add the sodium sulfite solution to the diluted sodium permanganate while stirring continuously.
- The reaction between the two compounds will produce manganese dioxide and sodium sulfate.
- Manganese dioxide is a harmless solid substance, while sodium sulfate is soluble in water.
Using Ascorbic Acid (Vitamin C)
Another reducing agent that can be used in disposing of sodium permanganate is ascorbic acid, also known as vitamin C. This method follows a similar process as using sodium sulfite:
- Dilute the sodium permanganate with water.
- Slowly add ascorbic acid to the diluted solution while stirring continuously.
- The reaction between ascorbic acid and sodium permanganate will produce carbon dioxide gas and manganese dioxide.
- Both of these substances are harmless and can be safely disposed of.
Precautions and Safety Measures for Disposing of Sodium Permanganate
Before you toss it in the trash or pour it down the drain, it’s crucial to understand the potential hazards involved in disposing of this chemical. As an expert on the topic, I have compiled some important precautions and safety measures to ensure a safe and proper disposal process.
First and foremost, handling sodium permanganate requires utmost care. This chemical is a strong oxidizing agent and can cause skin and eye irritation. If contact does occur, immediately rinse the affected area with plenty of water for at least 15 minutes. It is also recommended to wear protective gear such as gloves, safety glasses, and a face mask when handling large quantities.
Next, find a well-ventilated area to work in. Sodium permanganate can release harmful fumes, so it’s important to avoid inhaling them. And before you start disposing of the chemical, make sure to read the safety data sheet (SDS) provided by the manufacturer. This will provide detailed information on the proper handling and disposal methods specific to the product.
When disposing of sodium permanganate, keep it away from heat or flames as it can react violently when exposed to these sources. Also, be sure to keep it away from other chemicals or materials that may react with it. In case of a spill or accidental mixing, follow the appropriate steps outlined in the SDS.
Now that you’ve taken all necessary precautions, it’s time to dispose of sodium permanganate. The best method is to use reducing agents such as sodium sulfite or ascorbic acid. These substances neutralize the dangerous oxidizing properties of sodium permanganate and transform it into harmless compounds that can be easily disposed of.
Proper Disposal Procedures and Regulations
As a chemist, you know the importance of handling hazardous chemicals with care. And when it comes to disposing of them, the same level of caution and responsibility must be taken. One such chemical that requires proper disposal is sodium permanganate.
Sodium permanganate is commonly used in laboratories and industrial settings as a powerful oxidizing agent. However, it can also be a hazardous chemical if not handled and disposed of properly. In this guide, we will provide you with expert tips and insights on how to safely and responsibly dispose of sodium permanganate.
Check with Local Authorities or Waste Management Facilities
Before disposing of sodium permanganate, it is crucial to first consult with your local authorities or waste management facilities. Different regions may have specific regulations or guidelines for disposing of hazardous chemicals, so it is important to follow their instructions.
Chemical Neutralization or Incineration?
Generally, there are two main methods for disposing of sodium permanganate: chemical neutralization and incineration. Chemical neutralization involves mixing the chemical with another substance, such as an acid, to render it non-hazardous. On the other hand, incineration involves burning the chemical at high temperatures to break it down into less harmful compounds.
For small quantities of sodium permanganate, chemical neutralization is often the preferred method. However, it is important to use caution and follow proper safety protocols when handling acids. Incineration should only be done in licensed facilities that are equipped to handle hazardous waste.
Hazardous Waste Collection Programs
If handling and disposing of sodium permanganate yourself seems daunting, don’t worry – there are other options available. Many communities offer hazardous waste collection programs where residents can drop off their hazardous chemicals for proper disposal. This is a safe and convenient option for those who are not equipped to handle hazardous chemicals themselves.
Proper Storage is Key
Aside from following proper disposal procedures, it is also important to store sodium permanganate properly. The chemical should be kept in a cool, dry place away from any flammable materials and stored in its original container with a tight-fitting lid. This will help prevent any accidents or spills.
Conclusion
We have reached the end of our quest to safely dispose of sodium permanganate. This comprehensive guide has equipped you with all the necessary knowledge and precautions to ensure a responsible and seamless disposal process.
Despite its alluring purple hue, sodium permanganate can be a potential hazard if not handled with care. From skin irritation to environmental contamination, improper disposal can have serious consequences.
But worry not, for we have learned the proper steps such as dilution with water or neutralization with reducing agents, as well as taking necessary safety measures to bid farewell to this powerful chemical without any harm. And for those who are not equipped to handle hazardous chemicals themselves, there are always options such as consulting with professionals or utilizing hazardous waste collection programs.
Let us always remember that proper storage is crucial in preventing accidents and spills. So let’s handle them with utmost caution and responsibility, for we are chemists after all.
Thank you for embarking on this journey with us in uncovering the secrets of responsible disposal of sodium permanganate. Now go ahead and treat yourself to your favorite tunes – you deserve it.